What is a Breathalyzer?
A breathalyzer test is a device that is used for estimating blood alcohol content from a breath sample. Breathalyzers are primarily used by Police Officers on the road side to determine if an individual has been drinking or not.
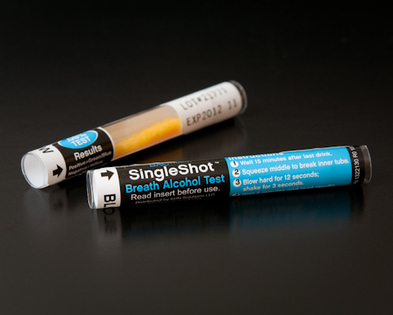
Initially there are two main parts to a breathalyzer. The first is the breathalyzer machine itself which is where the blood alcohol levels are displayed for the concluding results of the individual. Secondly, there is the disposable breathalyzer tester, which is where the individuals breathes into in order to determine the blood alcohol levels.
What are the chemical reactions that occur within the breathalyzer?
Breathalyzers are composed of potassium dichromate, sulfuric acid and silver nitrate in order to support the reactions with the ethanol, in determining the blood concentration levels in one’s body. When the potassium dichromate solution reacts with ethanol in the breathalyzer test, the potassium dichromate loses an oxygen atom. This process is well known to be called reduction.
Originally the potassium dichromate is orange, but once reacted with ethanol, the solution turns green, which now contains chromium sulfate. As this reduction process occurs, an oxidation process also occurs where the ethanol gets oxidized to acetic acid.
The occurrences of oxidation reactions after reduction reactions are commonly referred to as redox reactions.
Silver nitrate acts as a catalyst for the reaction in order to increase the rate at which the dichromate gets reduced during the reaction. Sulfuric acid in this breathalyzer test helps remove the alcohol from the exhaled air into the test solution, in order to provide the necessary acidic states.
The degree of colour change is related to the level of alcohol in the present air inside the chamber. To determine the amount of alcohol in the expelled air, the reacted mixture is compared to dishes of un-reacted mixture in the photocell system, which creates an electric current, which causes the needle inside the meter to move depending on the alcohol exposure levels.
Originally the potassium dichromate is orange, but once reacted with ethanol, the solution turns green, which now contains chromium sulfate. As this reduction process occurs, an oxidation process also occurs where the ethanol gets oxidized to acetic acid.
The occurrences of oxidation reactions after reduction reactions are commonly referred to as redox reactions.
Silver nitrate acts as a catalyst for the reaction in order to increase the rate at which the dichromate gets reduced during the reaction. Sulfuric acid in this breathalyzer test helps remove the alcohol from the exhaled air into the test solution, in order to provide the necessary acidic states.
The degree of colour change is related to the level of alcohol in the present air inside the chamber. To determine the amount of alcohol in the expelled air, the reacted mixture is compared to dishes of un-reacted mixture in the photocell system, which creates an electric current, which causes the needle inside the meter to move depending on the alcohol exposure levels.